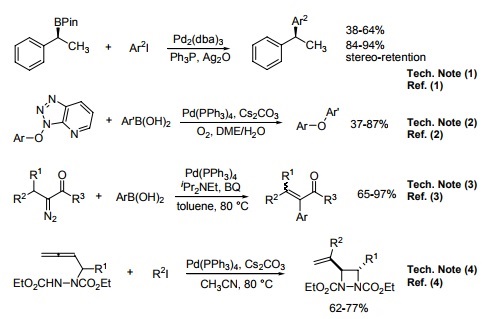
The stability of palladium(II) hydroxide and hydroxy–chloride complexes: an experimental solubility study at 25–85°C and 1 bar - ScienceDirect
Palladium(II) Chloride Complex Ion Recovery from Aqueous Solutions Using Adsorption on Activated Carbon

Solubility of palladium chloride benzonitrile complex in SC-CO 2 at (1)... | Download Scientific Diagram

The stability of palladium(II) hydroxide and hydroxy–chloride complexes: an experimental solubility study at 25–85°C and 1 bar - ScienceDirect

US20160340311A1 - Recovery and/or reuse of palladium catalyst after a suzuki coupling - Google Patents
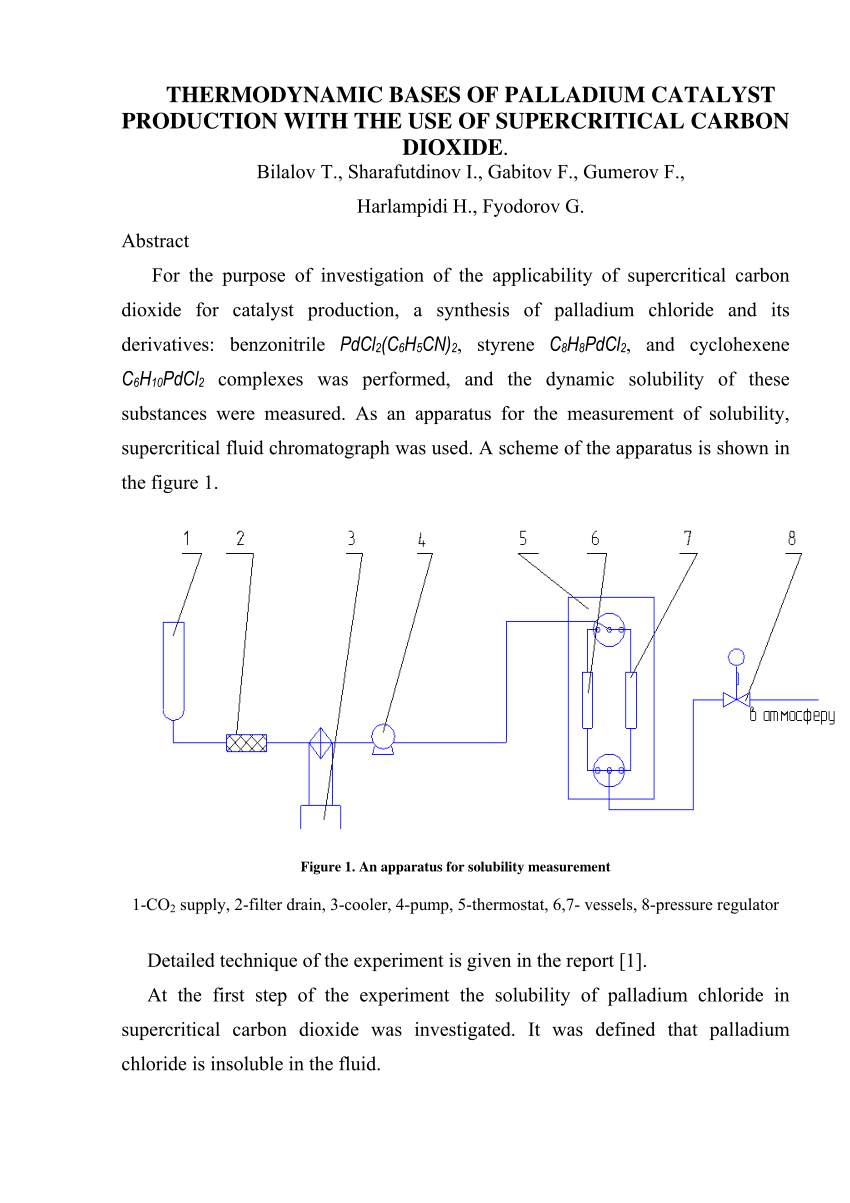
PDF) THERMODYNAMIC BASES OF PALLADIUM CATALYST PRODUCTION WITH THE USE OF SUPERCRITICAL CARBON DIOXIDE

Palladium Chloride at Rs 1200/gram | Palladium dichloride, Palladous chloride, पैलेडियम क्लोराइड - C. J. Chemicals Private Limited, Hyderabad | ID: 20756000555

The solubility of palladium in chloride solutions and the distribution of platinum, palladium, and related elements in hydrothermal mineralization